13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racem..

Solution For 13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g)
13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g) ₹ pressure. Then the ratio of the final volume to the initial volume of gases is:
Video solution 1: 13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g) ₹ pressure. Then the ratio of the final volume to the initial volume of gases is

1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25
Publicações Legais - Ilustrado
Solved 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15

Aquatics - Spring 2020 by Portland Parks & Recreation - Issuu
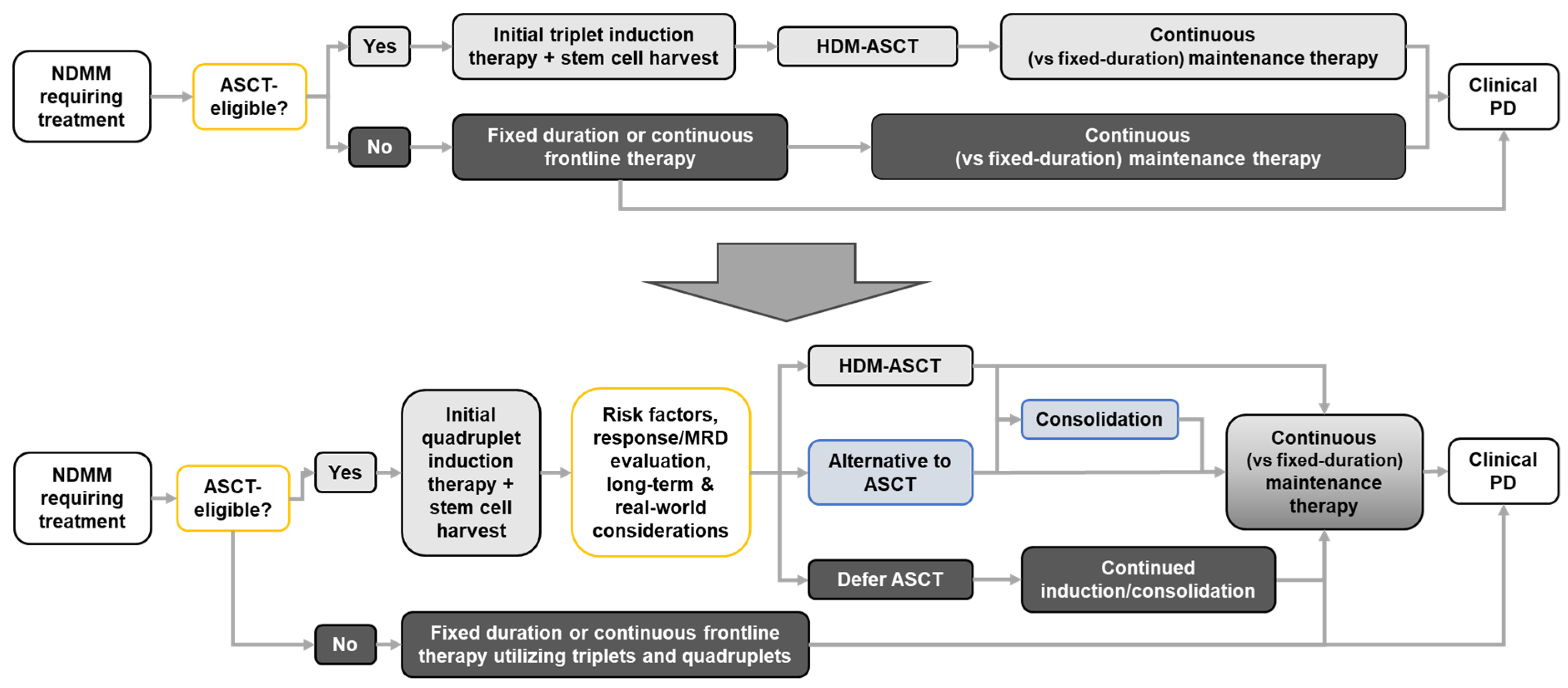
Cancers, Free Full-Text

JPM, Free Full-Text

Counting ppt download

2020 United States Senate election in Iowa - Wikipedia

Target shooting by hunters and their use of shooting ranges: 1975, 1991, and 2011. Addendum to the 2011 National Survey of Fishing, Hunting, and Wildlife-Associated Recreation - Documents - USFWS National Digital Library

Shopee Brasil Ofertas incríveis. Melhores preços do mercado
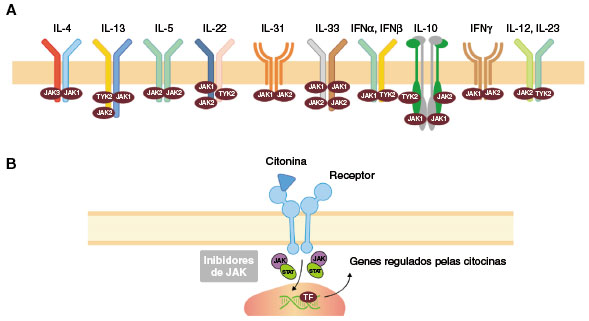
Arquivos de Asma, Alergia e Imunologia - AAAI

2020 integrated report of CEMEX, S.A.B. de C.V.

Solved Q4. OUTPUT: 12345 6 7 8 9 10 11 12 13 14 15 16 17 18

Socioeconomic variation in the financial consequences of ill

LEGAL SERVICES CORPORATION Board of Directors Meeting






