Cells, Free Full-Text
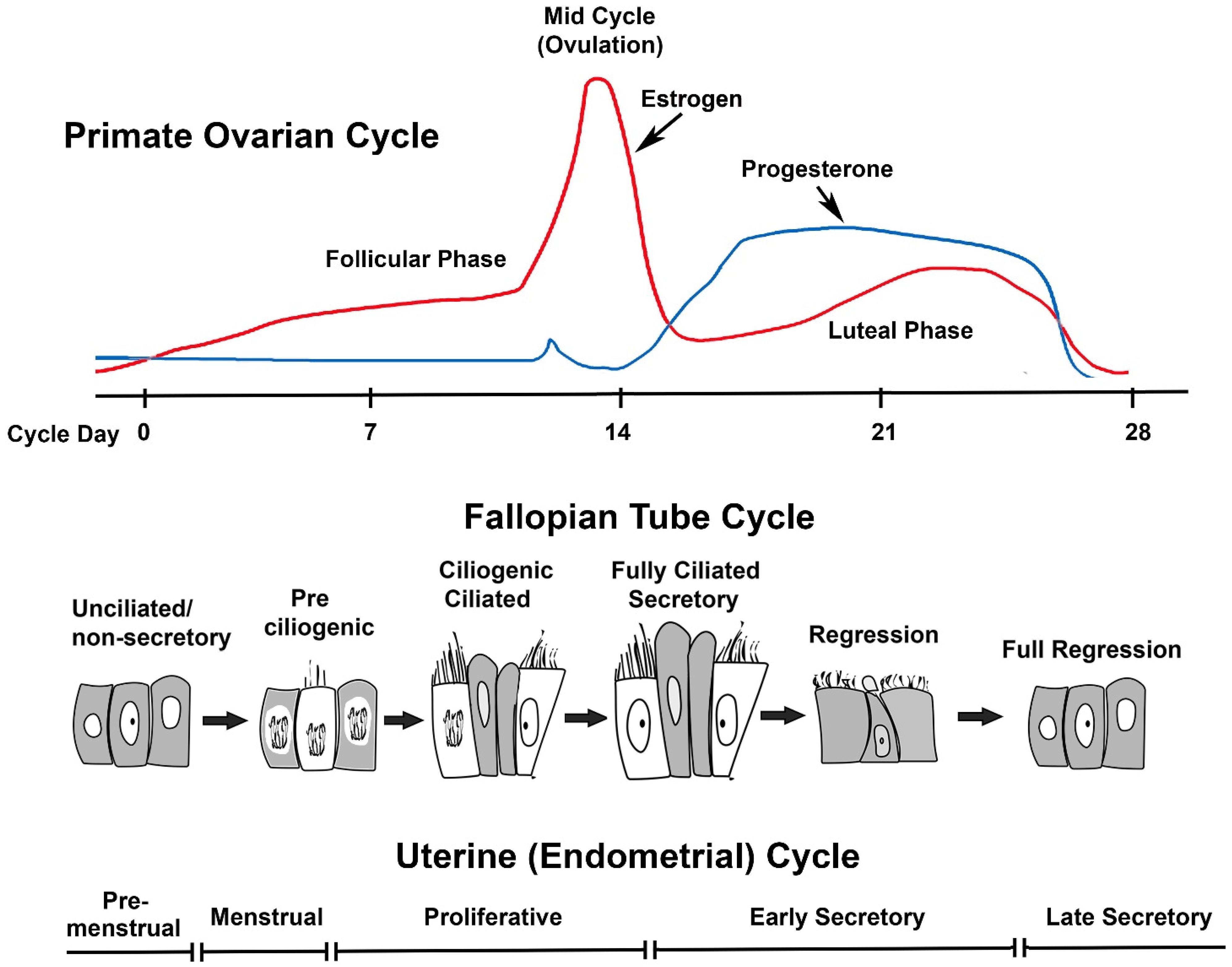
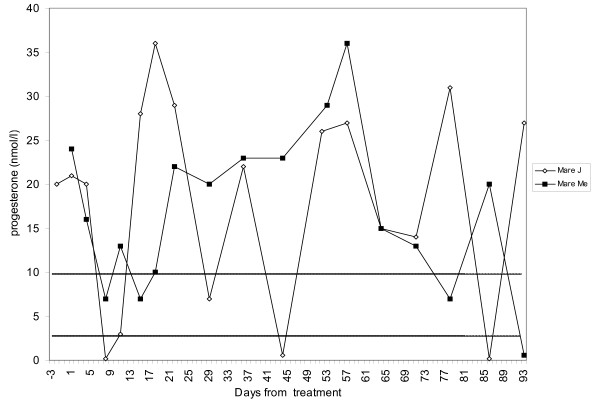
Therapies that target progesterone action hold potential as contraceptives and in managing gynecological disorders. Recent literature reviews describe the role of steroid hormones in regulating the mammalian oviduct and document that estrogen is required to stimulate epithelial differentiation into a fully functional ciliated and secretory state. However, these reviews do not specifically address progesterone action in nonhuman primates (NHPs). Primates differ from most other mammals in that estrogen levels are >50 pg/mL during the entire menstrual cycle, except for a brief decline immediately preceding menstruation. Progesterone secreted in the luteal phase suppresses oviductal ciliation and secretion; at the end of the menstrual cycle, the drop in progesterone triggers renewed estrogen-driven tubal cell proliferation ciliation secretory activity. Thus, progesterone, not estrogen, drives fallopian tube cycles. Specific receptors mediate these actions of progesterone, and synthetic progesterone receptor modulators (PRMs) disrupt the normal cyclic regulation of the tube, significantly altering steroid receptor expression, cilia abundance, cilia beat frequency, and the tubal secretory milieu. Addressing the role of progesterone in the NHP oviduct is a critical step in advancing PRMs as pharmaceutical therapies.

Cell Biology, 3rd Edition

Cell-free synthetic biology as an emerging biotechnology
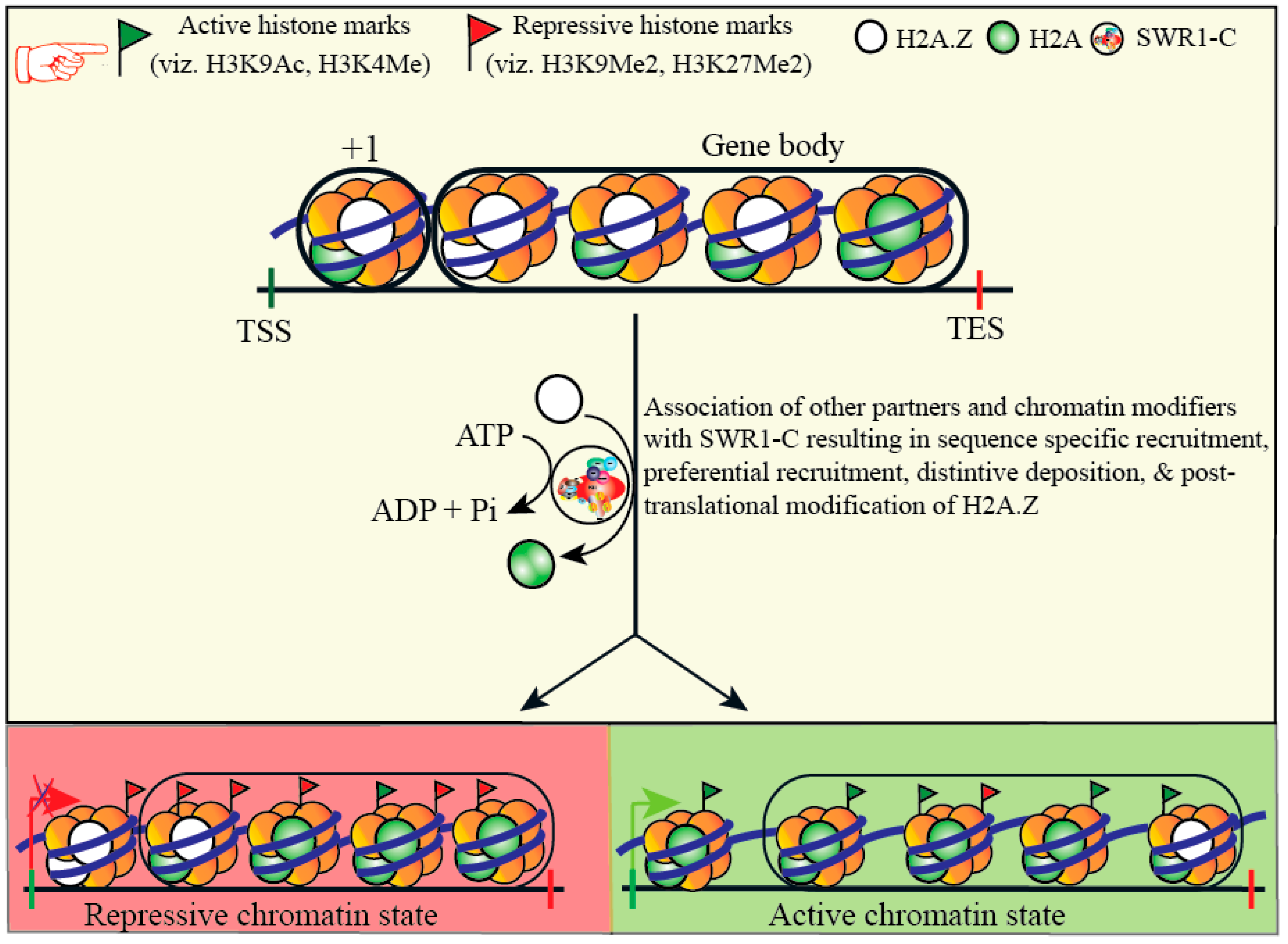
ChIP analysis of histone modifications at the Ig e locus in CL-01 cells, histone masque

Cells An Open Access Journal from MDPI

Cell Circuits and Complex Tissues

Cell-free Fetal DNA — A Trigger for Parturition

Cell number and cell density in the cerebellar cortex of man and
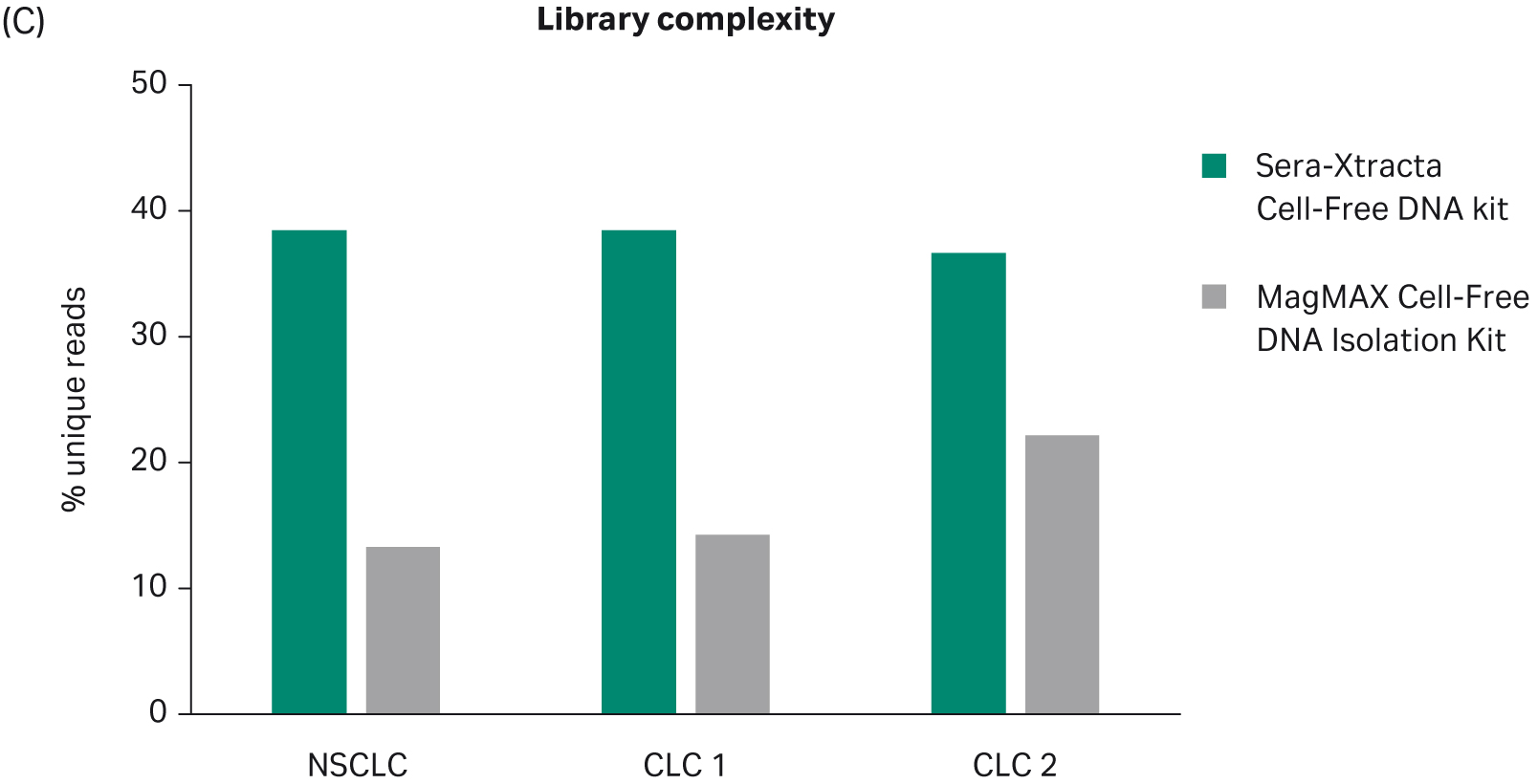
Discovery of cancer biomarkers in serum using Sera-Xtracta Cell
Cells, Free Full-Text

How to add text to the beginning or end of all cells in Excel?
Cells, Free Full-Text
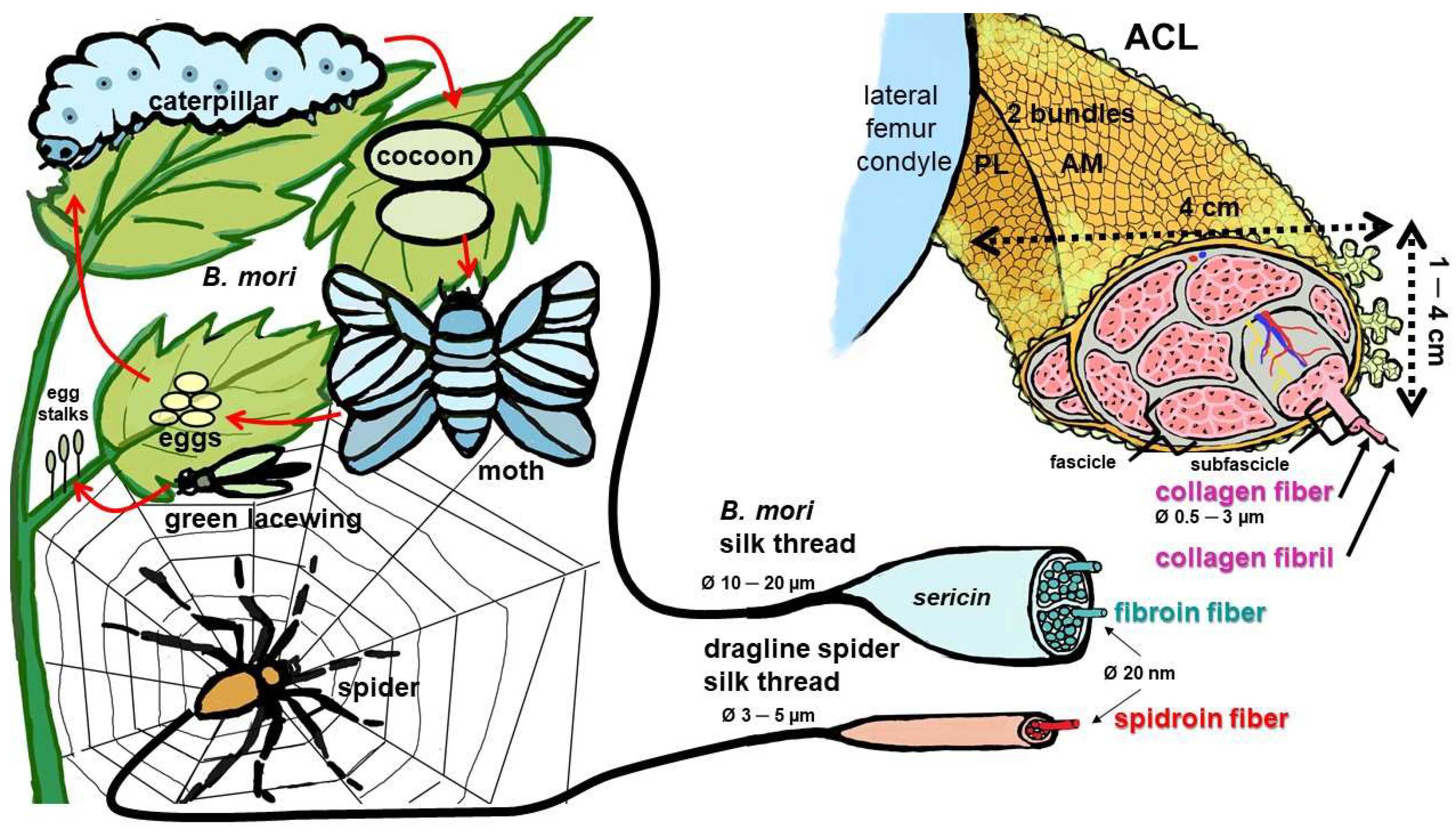
Cells, Free Full-Text, spider silk tibia
THE LIVES OF A CELL : LEWIS THOMAS : Free Download, Borrow, and
Cells, Free Full-Text, freecell online 100