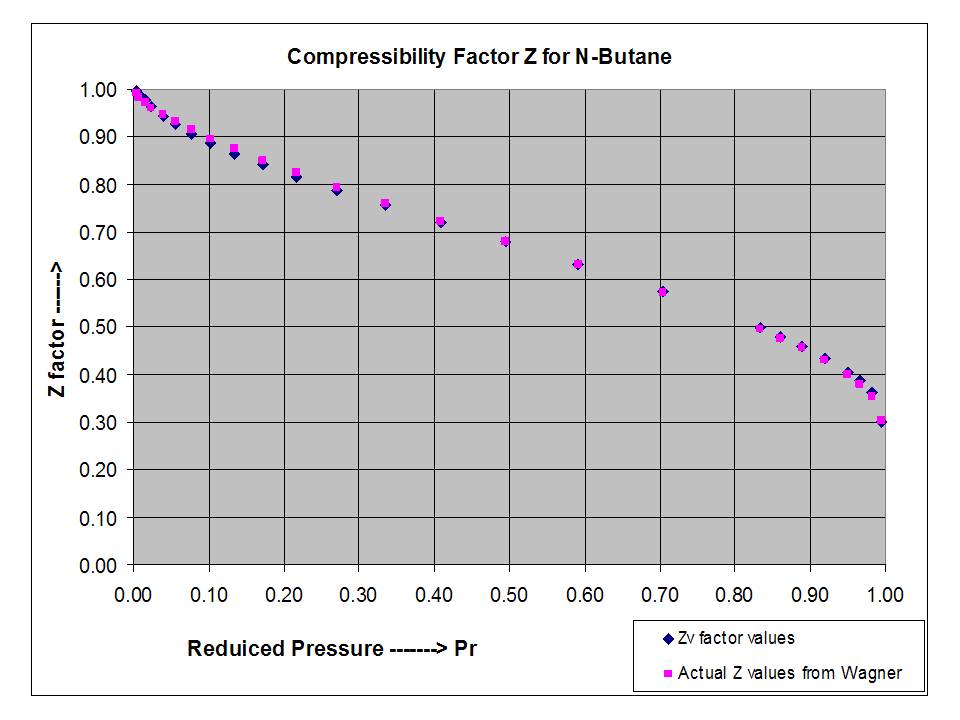
Compressibility Factor, z vs Pressure, P (kPa)
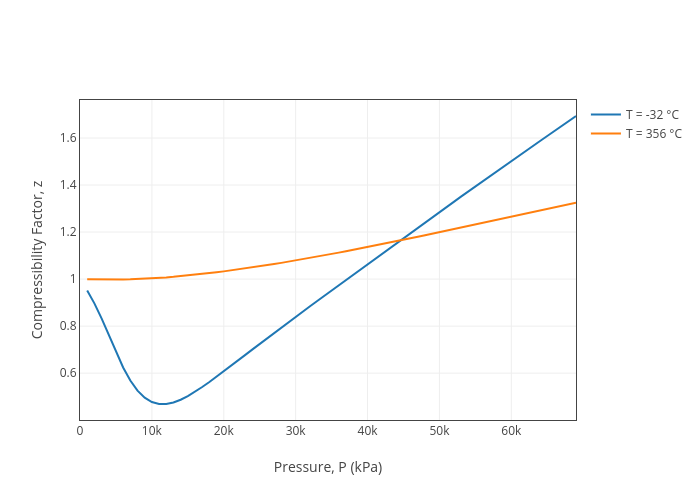
Jdvani's interactive graph and data of "Compressibility Factor, z vs Pressure, P (kPa)" is a line chart, showing T = -32 °C vs T = 356 °C; with Pressure, P (kPa) in the x-axis and Compressibility Factor, z in the y-axis..
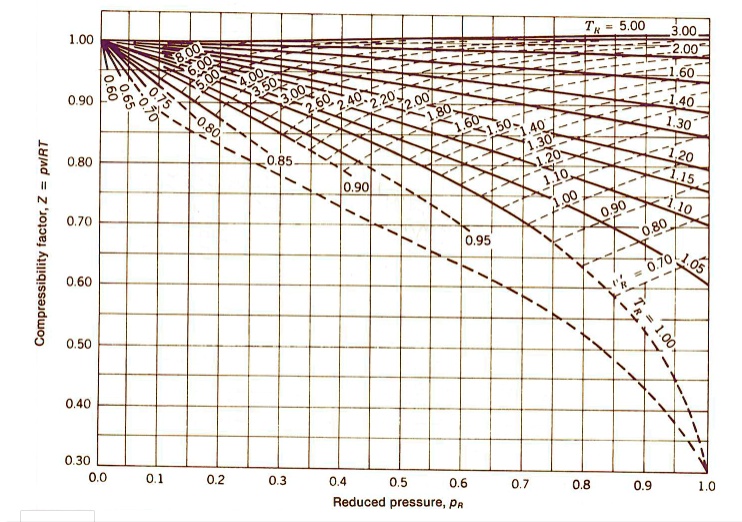
SOLVED: Determine the specific volume (m^3/kg) for air at T = -120.2°C and P = 57.2 bar using the generalized compressibility chart given below. Note: Give your answer to five decimal places.
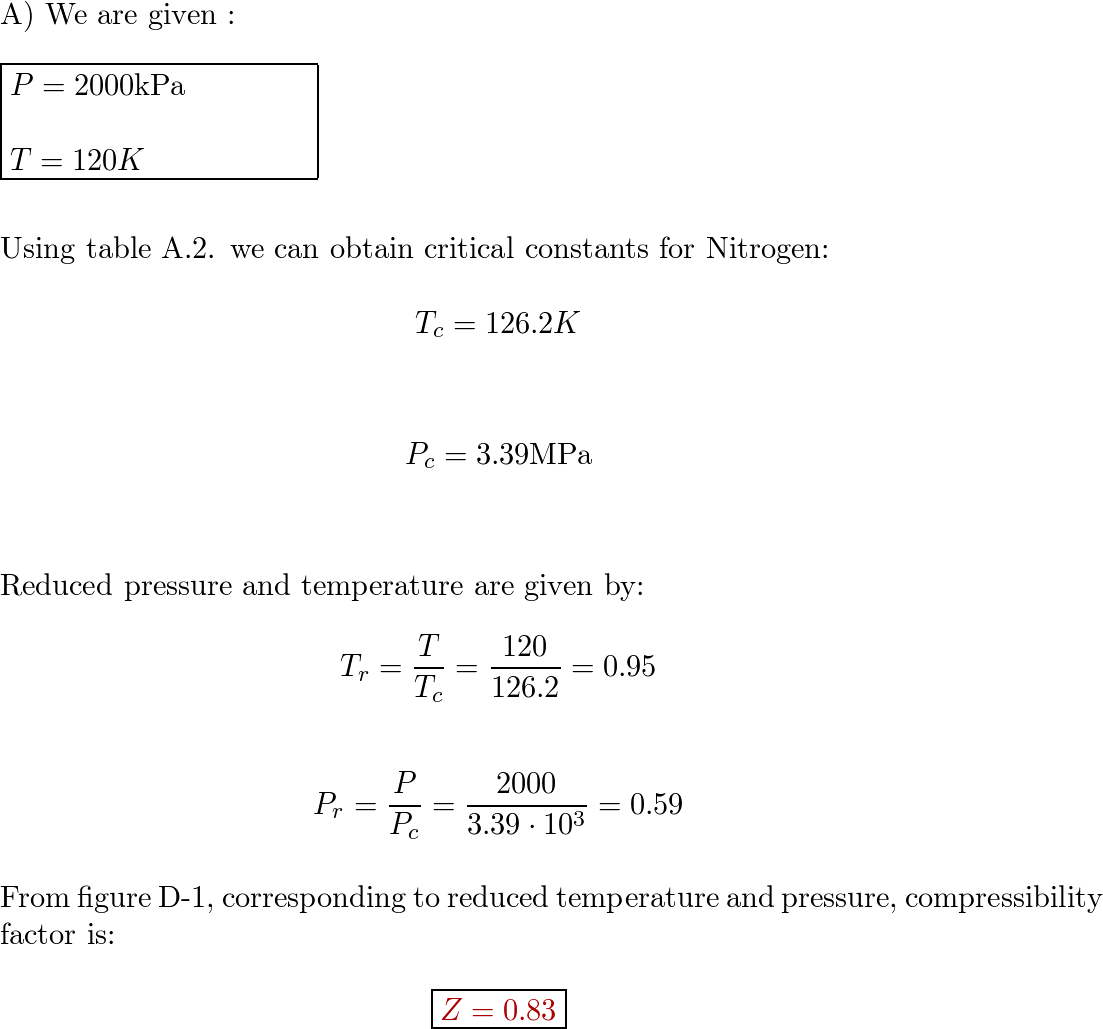
Find the compressibility factor for nitrogen at. 2000 kPa, 1

Compressibility Factor - an overview

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics
Standing and Kats Compressibility Factor Chart (Ahmed 2006)
How can a gas be ideal at a high pressure and low temperature? - Quora

Short handy Formulas calculating Densities and Enthalpies of Saturated Steam

How accurate (find $Z$ ) is it to assume that propane is an
Compressibility factor of water

Reading Compressibility Factor Charts

Compressibility of Liquids - an overview

physical chemistry - Compressibility Factor Graph - Which gas attains a deeper minimum? - Chemistry Stack Exchange
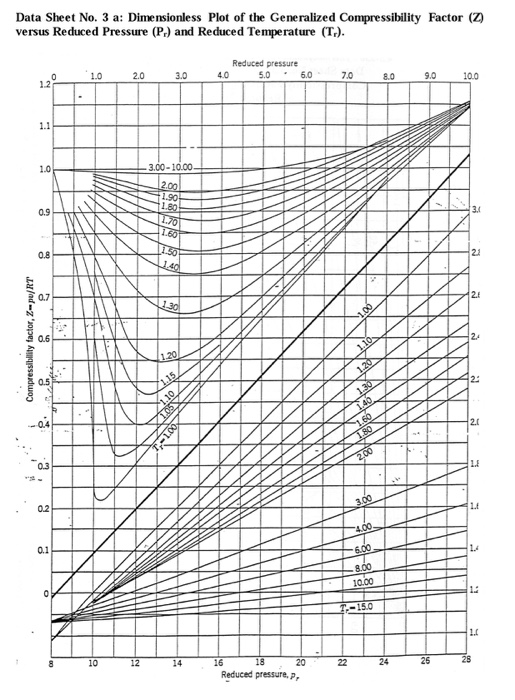
Solved I have some propane gas at a pressure 7 MPa and a
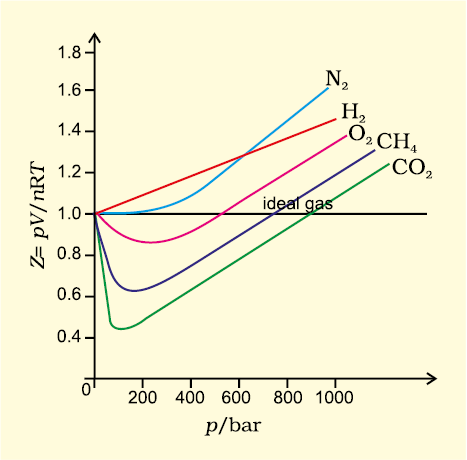
Sections

Air Compressibility Factor Table - EnggCyclopedia