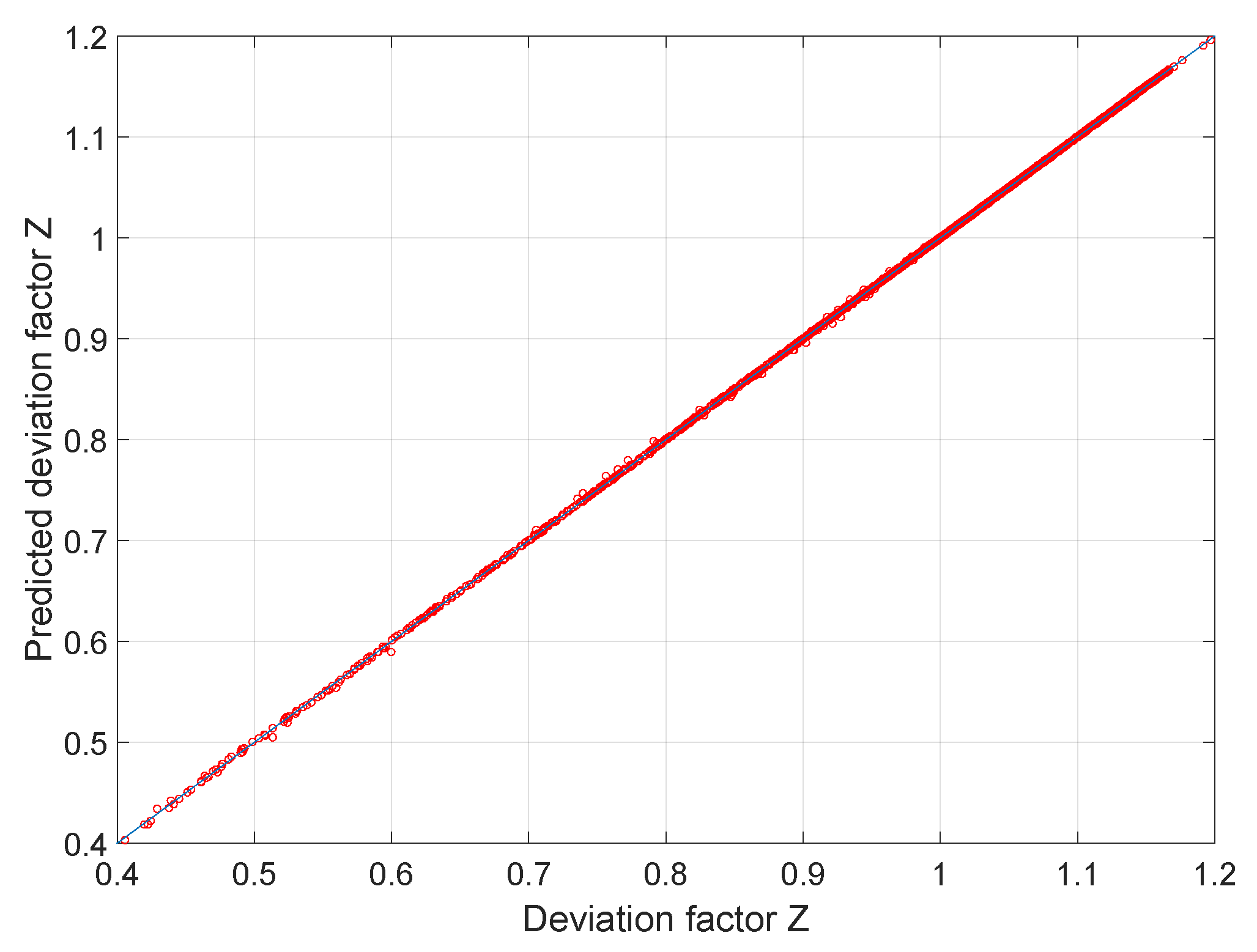
Compressibility factor Z - Gaseous State


Solved - Evaluate the compressibility factor of two

EXERCISE States of Matte pressure and volume. The compressibility

Math cad compressibility factor, z, of real gas using the redlich
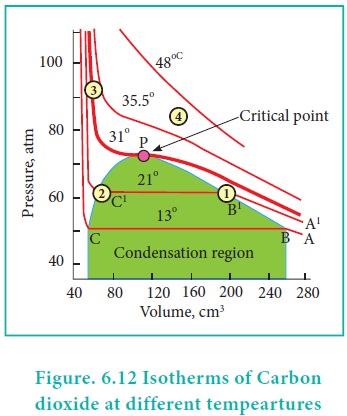
Pressure-Volume isotherms of Carbon dioxide
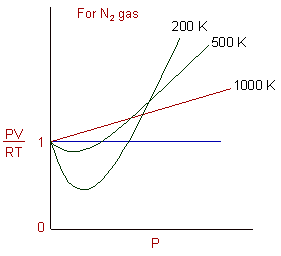
Ideal gases and real gases are compressible or not compressible what is the compressible factor for real gases and ideal gases.

Compressibility Factor Z

Propane compressibility factors.
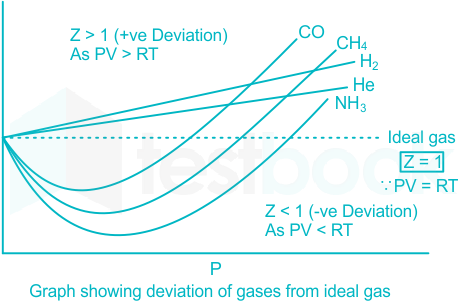
REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR

Compressibility factor, Z of a gas is given as Z = pV / nRTi What
Solved] The compressibility factor for an ideal gas is

What is Compressibility Factor (Z)?, Gaseous state, JEE/NEET

Compressibility factor Z, gaseous state Class 11th
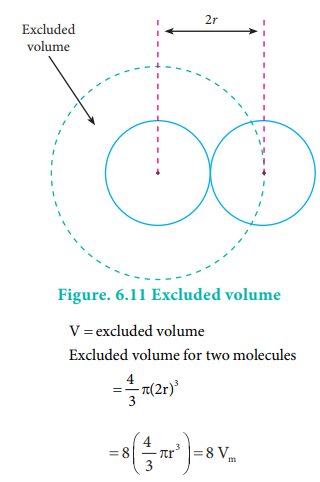
Deviation from ideal gas behaviour
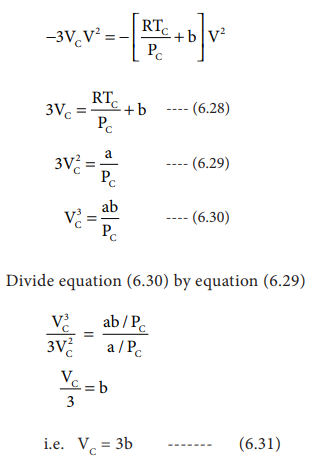
Derivation of critical constants from van der Waals constant

Graham' s Law of Diffusion