In the following compressibility factor Z vs pressure graph at 300

In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state

Modus Operandi for Practice: A Comprehensive Study Package on Atomic Structure and the Gaseous State, PDF, Spectroscopy

Compressibility factor (gases) - Knowino

47. In the following compressibility factor (Z) vs pressure graph 300 K, the compressibility factor of CH4 pressures < 200 bar deviates from ideal behavior because
For a real gas at 25°C temperature and high pressure (99 bar) the value of compressibility factor is 2, - Sarthaks eConnect
47. In the following compressibility factor (Z) vs pressure graph 300 K, the compressibility factor of CH4 pressures < 200 bar deviates from ideal behavior because

Modus Operandi for Practice: A Comprehensive Study Package on Atomic Structure and the Gaseous State, PDF, Spectroscopy
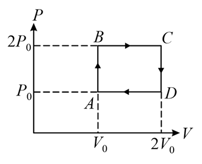
What is the expression of first law of thermodynamics for adiabatic process

2nd Year Holiday Assignment, PDF, Tangent

Gas Laws / Gases Behaviour, PDF, Gases

2nd Year Holiday Assignment, PDF, Tangent