Molecules, Free Full-Text
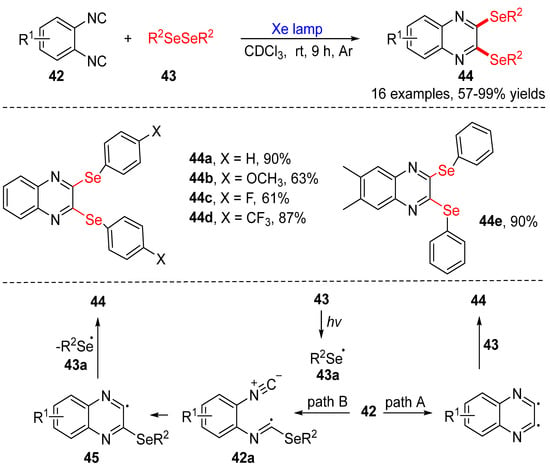
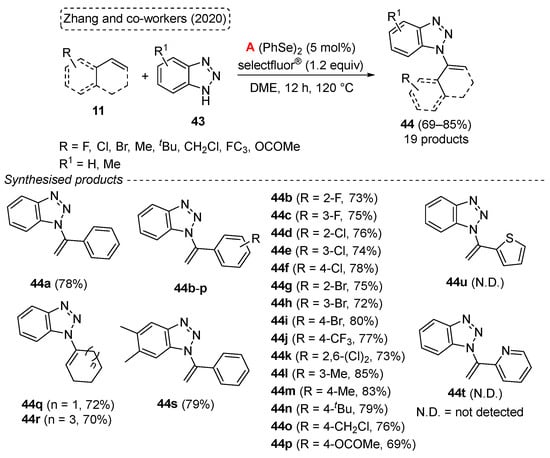
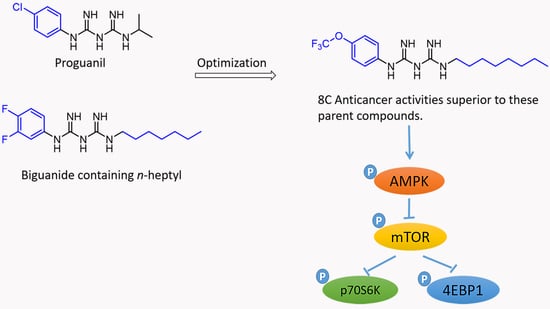
Dichalcogenides (disulfides and diselenides), as reactants for organic transformations, are important and widely used because of their potential to react with nucleophiles, electrophilic reagents, and radical precursors. In recent years, in combination with photochemical technology, the application of dichalcogenides as stable radical reagents has opened up a new route to the synthesis of various sulfur- and selenium-containing compounds. In this paper, synthetic strategies for disulfides and diselenides and their applications with photochemical technology are reviewed: (i) Cyclization of dichalcogenides with alkenes and alkynes; (ii) direct selenylation/sulfuration of C−H/C−C/C−N bonds; (iii) visible-light-enabled seleno- and sulfur-bifunctionalization of alkenes/alkynes; and (iv) Direct construction of the C(sp)–S bond. In addition, the scopes, limitations, and mechanisms of some reactions are also described.
Molecules, Free Full-Text, chinese wall cpa 20
Molecules, Free Full-Text, chinese wall cpa 20
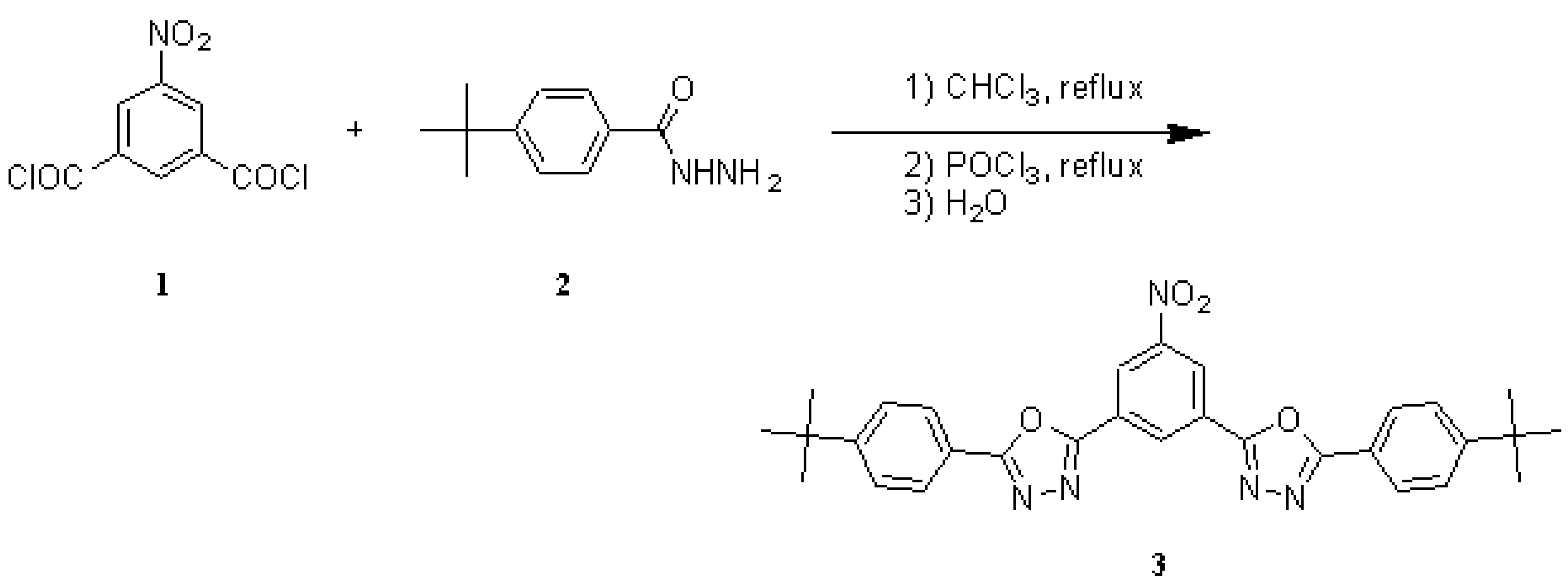
Molecules, Free Full-Text

Molecules, Free Full-Text, players max cigarettes where to buy
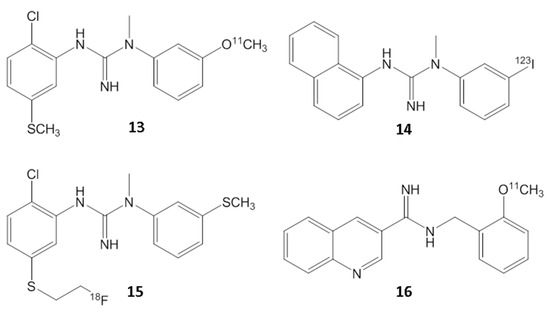
Molecules, Free Full-Text, fotos de menin

Molecules, Free Full-Text, chinese wall cpa 20

Molecules, Free Full-Text, Stearic Acid
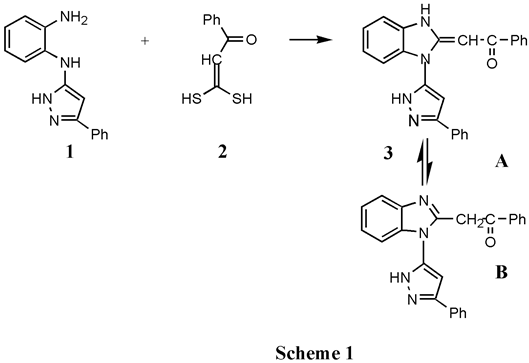
Molecules, Free Full-Text
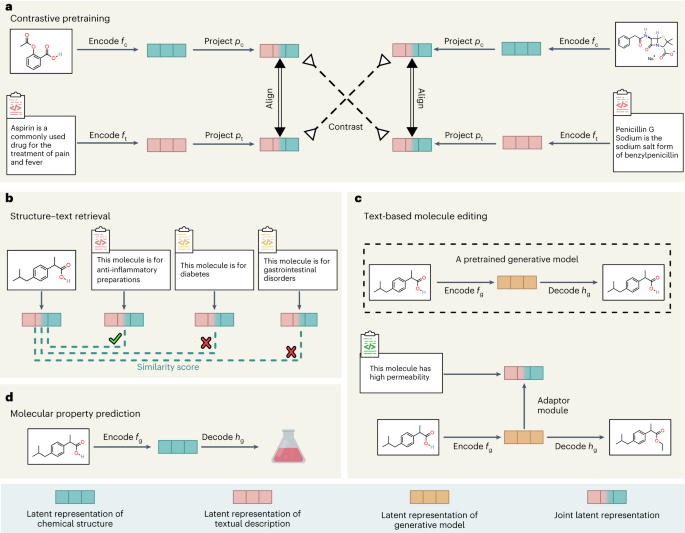
Molecules, Free Full-Text, kelly godoy coelho