
The NGAL Test is a particle-enhanced turbidimetric immunoassay for the quantitative determination of NGAL in human urine and plasma on automated clinical chemistry analyzers. NGAL measurements are useful in the risk assesment of AKI.

NGAL - Bioporto

Elevated Neutrophil Gelatinase-Associated Lipocalin Is Associated With the Severity of Kidney Injury and Poor Prognosis of Patients With COVID-19 - ScienceDirect

News - Bioporto

About BioPorto - Bioporto

The Prognostic Utility of Plasma NGAL Levels in ST Segment Elevation in Myocardial Infarction Patients
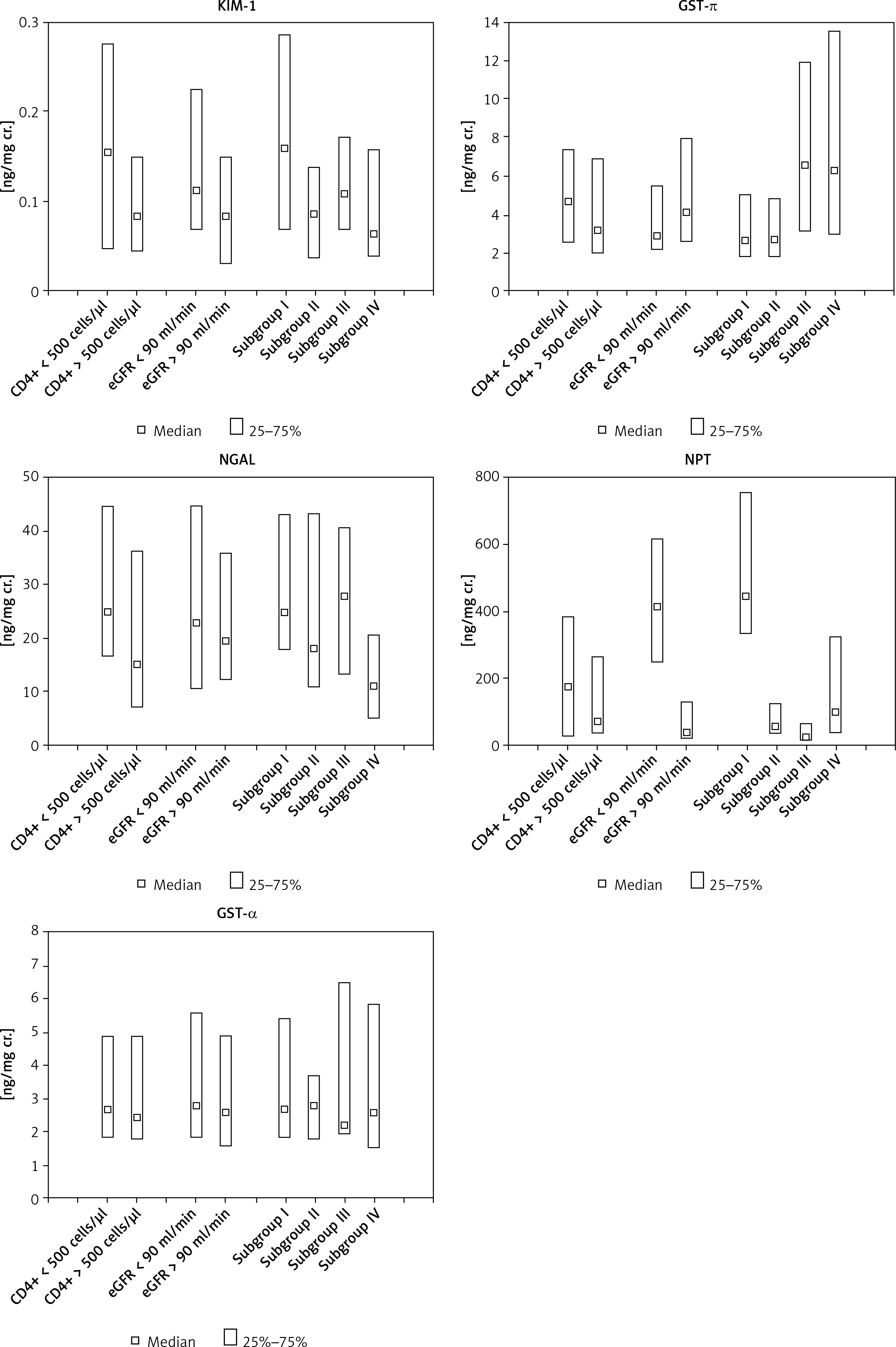
Selected biochemical parameters in the urine of HIV-infected patients in monitoring of kidney function
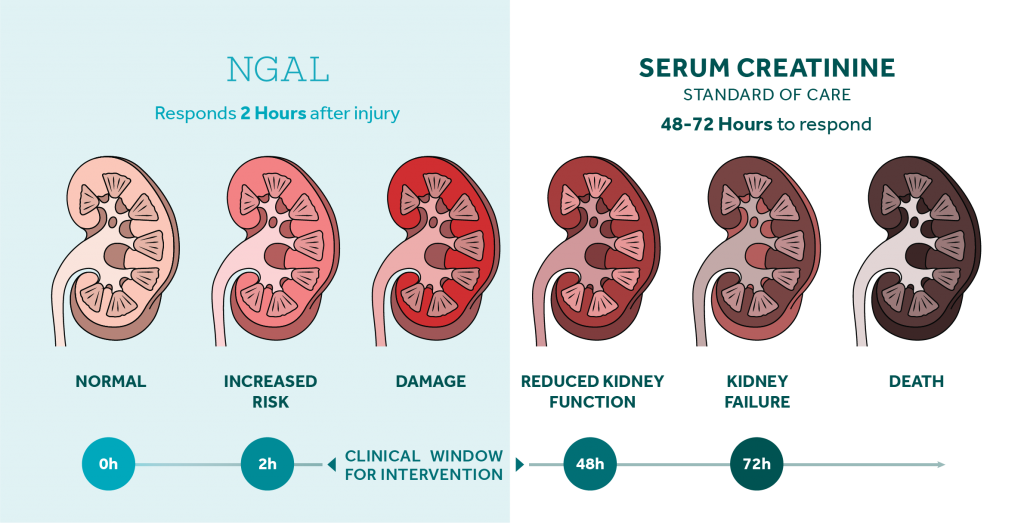
NGAL - Bioporto
/wp-content/uploads/2023/12/ProNephro
BioPorto Submits Application for Marketing Authorization of NGAL Test to the US Food and Drug Administration

About BioPorto - Bioporto

PDF) Does NGAL reduce costs? A cost analysis of urine NGAL (uNGAL) & serum creatinine (sCr) for acute kidney injury (AKI) diagnosis

BioPorto Diagnostics A/S: Contact Details and Business Profile

NGAL - Bioporto






