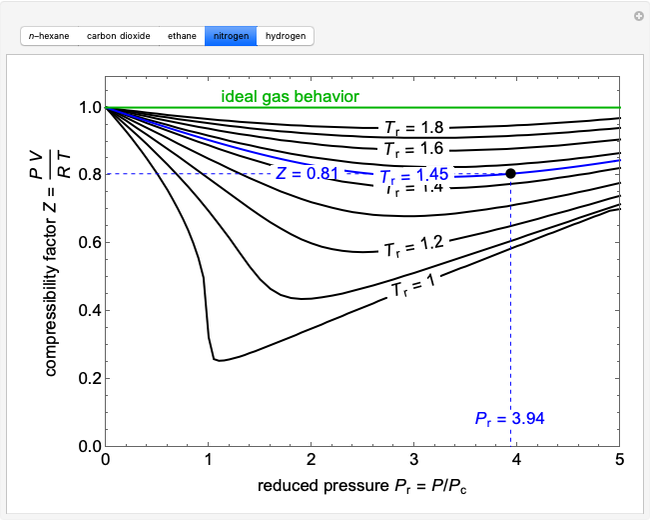
The compressibility factor Z a low-pressure range of all gases except hydrogen is:Z=(1+ displaystylefrac{a}{V_{m}RT})Z =(1-displaystylefrac{a}{V_{m}RT})Z=(1+displaystylefrac{Pb}{RT})Z = ( 1 - displaystylefrac{Pb}{RT})

Click here:point_up_2:to get an answer to your question :writing_hand:the compressibility factor z at a lowpressure range of all gases except hydrogen is
Click here👆to get an answer to your question ✍️ The compressibility factor Z a low-pressure range of all gases except hydrogen is-Z-1- displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-Pb-RT-Z - - 1 - displaystylefrac-Pb-RT-
The van der Waals equation for real gases is -P-aVm2-Vm-x2212-b-RT

Numerical simulation of fractured horizontal well considering threshold pressure gradient, non‐Darcy flow, and stress sensitivity - Xiao - 2023 - Energy Science & Engineering - Wiley Online Library

Except H_2 and He, the compressibility factor Zleft [ =frac{PV}{nRT } right ]< 1 all gases low pressure.

Compressibility factor - Wikipedia

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

Gas Compressibility - an overview

1402.1205] Thermodynamic Properties of the van der Waals Fluid

Gas compressibility factor Z: Ideal gas vs Real gas

Non-Ideal Gas Behavior Chemistry: Atoms First

Non-Ideal Gas Behavior Chemistry: Atoms First