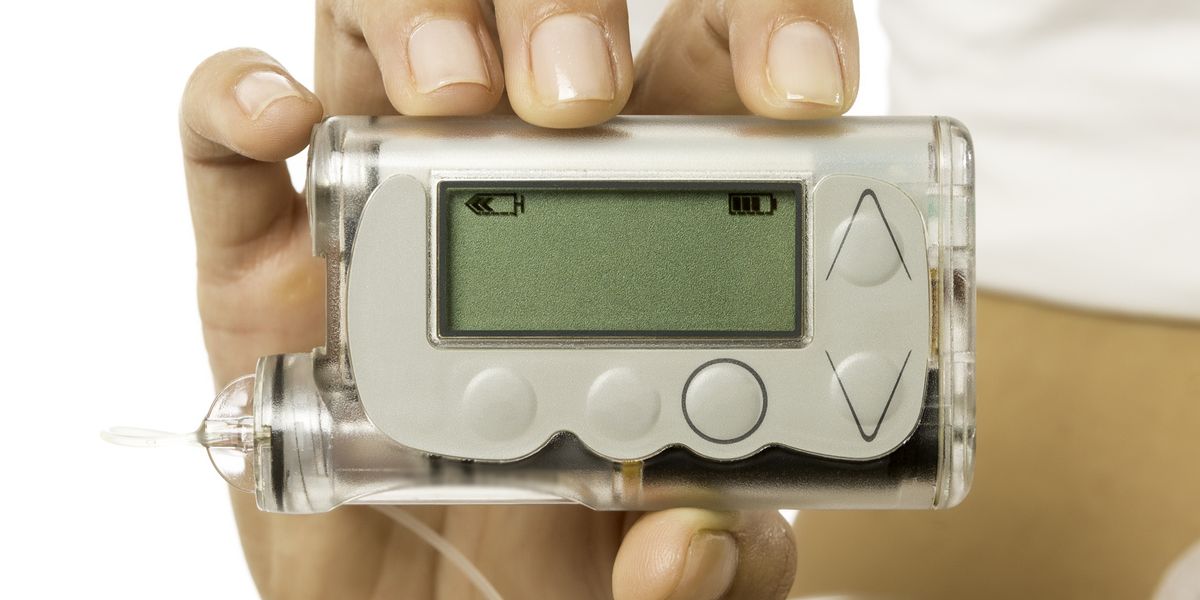
PharmaSens seeks FDA approval for new insulin pump
PharmaSens has submitted an application to obtain FDA approval for its ‘niia essential’ insulin patch pump system.

Report: Medtronic in negotiations to buy Israel-based insulin pump

FDA Advisory No.2023-0589

PharmaSens AG on LinkedIn: #pharmasensinnovation #attd2024

Metabolic Disorders Archives - Medical Device Network

Regulation Archives - Medical Device Network

Medical Device Network

Diabetotech

FDA Advisory No. 2020-1466
Regulation Archives - Medical Device Network

Animas® Vibe™ Insulin Pump and Continuous Glucose Monitoring

Kazakhstan Archives - Medical Device Network

FDA warns of cybersecurity risk with certain Medtronic insulin pumps

The Roche Accu-Chek Solo micropump – so just what is it

Thoughts from Shore, including paper injection moulding, an FDA

Insulet warns of battery issues with some Omnipod Dash devices