Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in

Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to T 2. The final pressure Pf is :P i T 1 T 2/ T 1+ T 2B. 2 P i T 1/ T 1+ T 2C. 2 P i T 1 T 2/ T 1+ T 2D. 2 P i T 2/ T 1+ T 2
Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in the figure below- The temperature of one of the bulbs is then raised to T 2- The final pressure Pf is -P i T 1 T 2- T 1- T 2B- 2 P i T 1- T 1- T 2C- 2 P i T 1 T 2- T 1- T 2D- 2 P i T 2- T 1- T 2
The correct option is D 2P_i ( T_2T_1+T_2 )Since the above system is a closed one, the total number of moles of the ideal gas will be equal before and after th

An ideal gas at a pressure of 152 kPa is contained in a bulb of u
Two flasks at the same temperature are joined by a glass tube with a stopcock. Flask A is a 4.0 L flask containing N2 (g) at 2.0 ATM, while flask B is
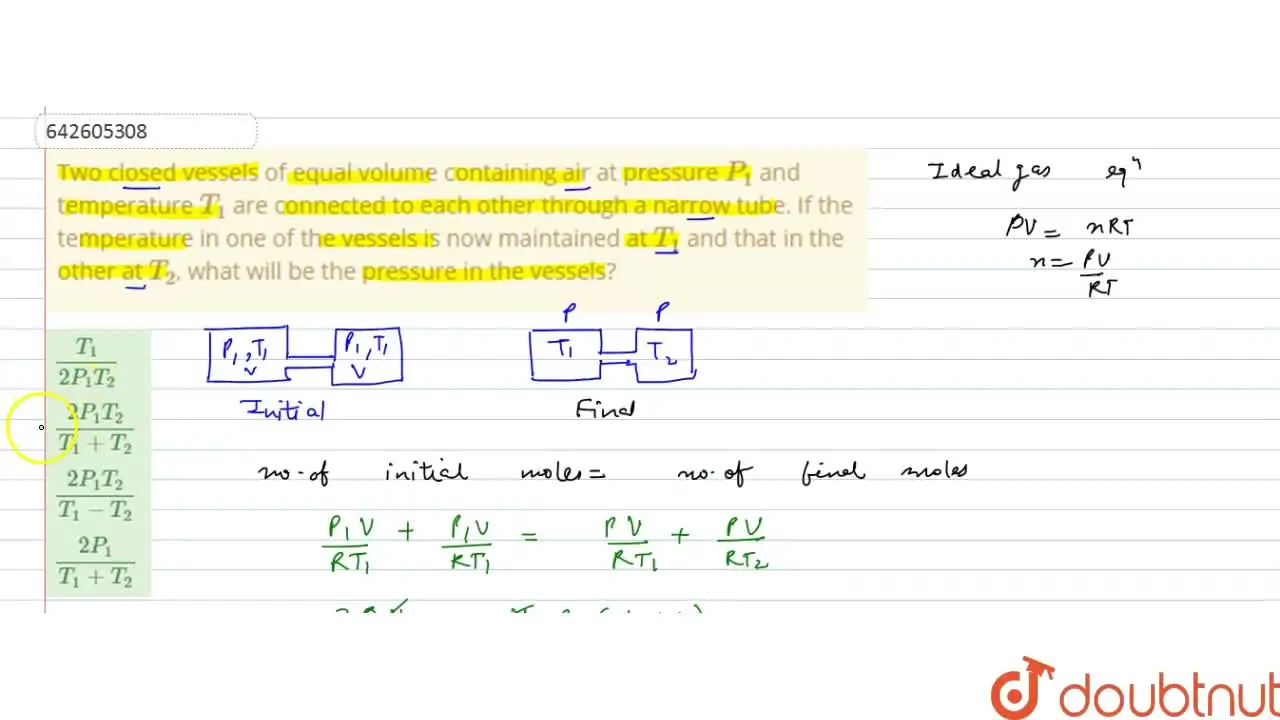
Two closed vessels of equal volume containing air at pressure P(1) and
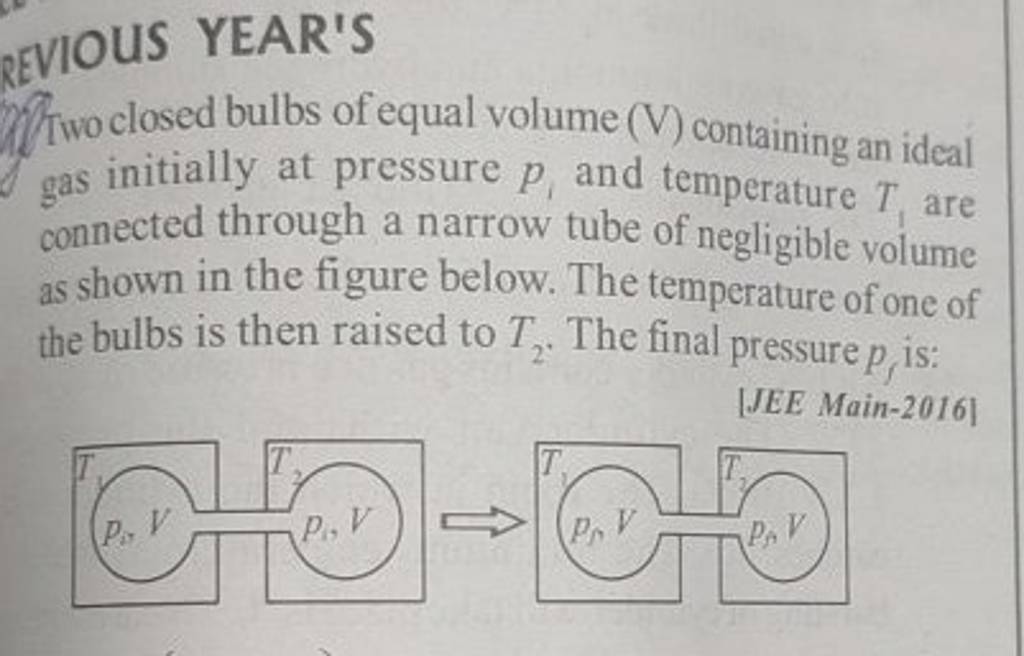
Two closed bulbs of equal volume ( V ) containing an ideal gas initially ..

Two closed bulbs of equal volume (V) containing an ideal gas initially pressure p and temperature T. are connected through a narrow tube of negligible volume as shown in the figure below.

⏩SOLVED:Two closed bulbs of equal volume (V) containing an ideal gas…

Vipin Singh - student of IIT ROORKEE - BHU and Now in IIT - EduRev

Two closed bulbs of equal volume V containing an ideal gas initially at pressure P 1 and temperature T 1 are connected through a narrow tube of negligible volume as shown in

Solved A 2. Two identical glass bulbs are connected by a
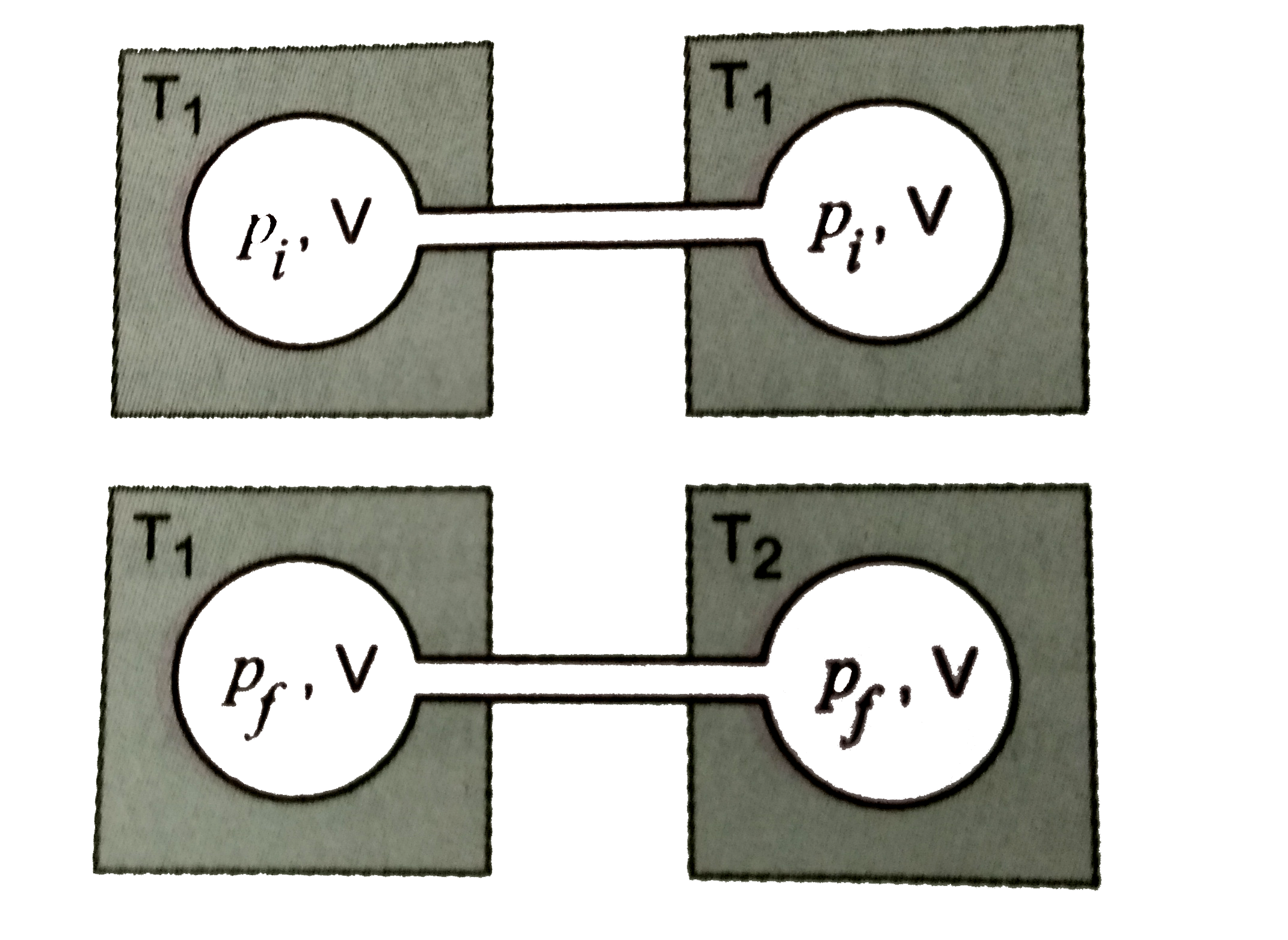
Two closed bulbs of equal volume (V) containing an ideal gas initially

TU Tiso dosed bulbs of equal volume s initially pressure p and











