
The present review gives an overview over non-toxic cyanation agents and cyanide sources used in the synthesis of structurally diverse products containing the nitrile function. Nucleophilic as well as electrophilic agents/systems that transfer the entire CN-group were taken in consideration. Reactions in whi

Reductive cyanation of organic chlorides using CO2 and NH3 via Triphos–Ni(I) species

Copper-Mediated Transformation of Organosilanes to Nitriles with

Optimization of Reaction Conditions for Reduction of Benzoyl Cyanide

11C-, 12C-, and 13C-cyanation of electron-rich arenes via organic photoredox catalysis - ScienceDirect

Molecules, Free Full-Text

Non-toxic cyanide sources and cyanating agents - Organic

Fabrication of Pd Nanoparticles Embedded C@Fe3O4 Core–Shell Hybrid
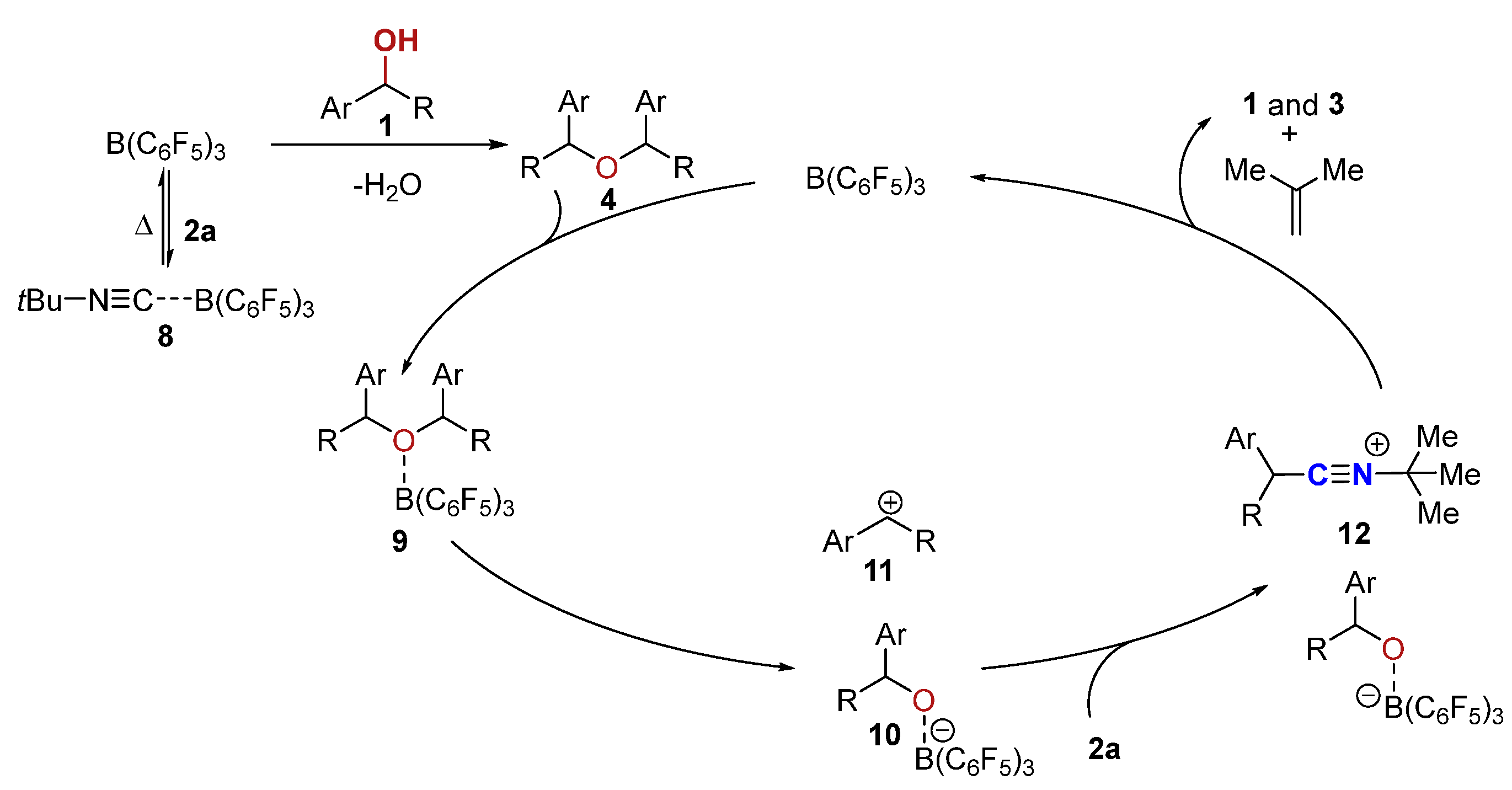
Molecules, Free Full-Text

PDF] Cu-Catalyzed direct cyanation of terminal alkynes with AMBN or AIBN as the cyanation reagent.

PDF) Reductive cyanation of organic chlorides using CO2 and NH3 via Triphos–Ni(I) species

Light-Triggered, Ni-Catalyzed Cyanation of Aryl Triflates with 1,4-Dicyanobenzene as the CN Source

氰化氢- 维基百科,自由的百科全书

Copper-catalyzed direct cyanation of terminal alkynes with benzoyl cyanide - ScienceDirect





