A new catalyst can make ethanol out of carbon dioxide
Scientists from Argonne National Laboratory, the University of Chicago’s Pritzker School of Molecular Engineering and Northern Illinois University have helped discover a new electrocatalyst that can consistently convert carbon dioxide and water into ethanol. That means the carbon dioxide emitted from industrial processes—such as fossil fuel or alcohol fermentation plants—can be turned into a valuable commodity at reasonable cost.
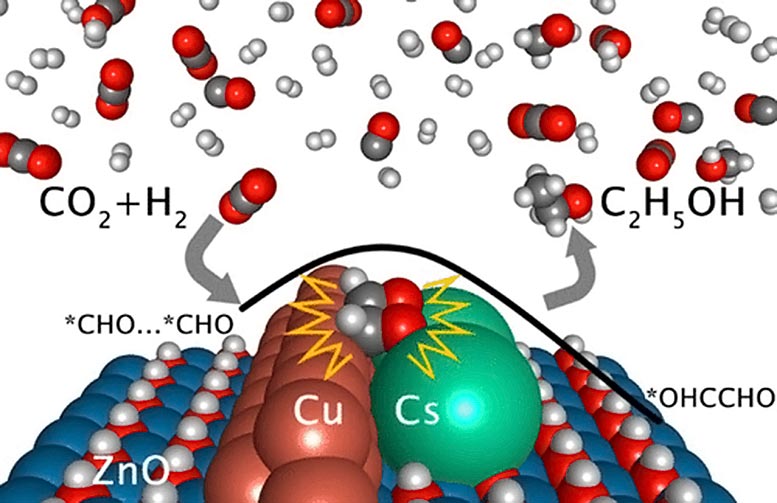
Three-Part Catalyst Helps Transform Excess CO2 Into Usable Ethanol
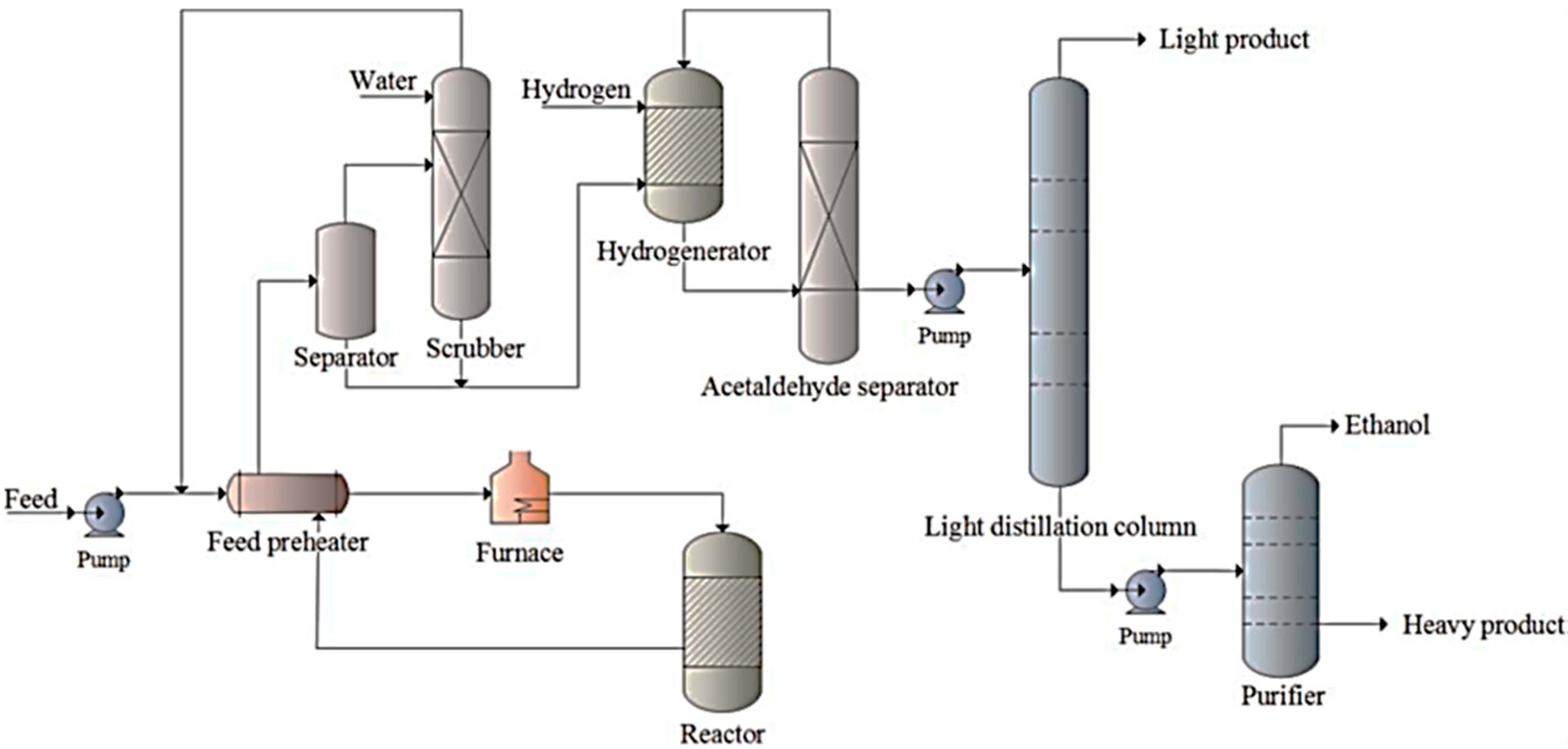
Catalysts, Free Full-Text
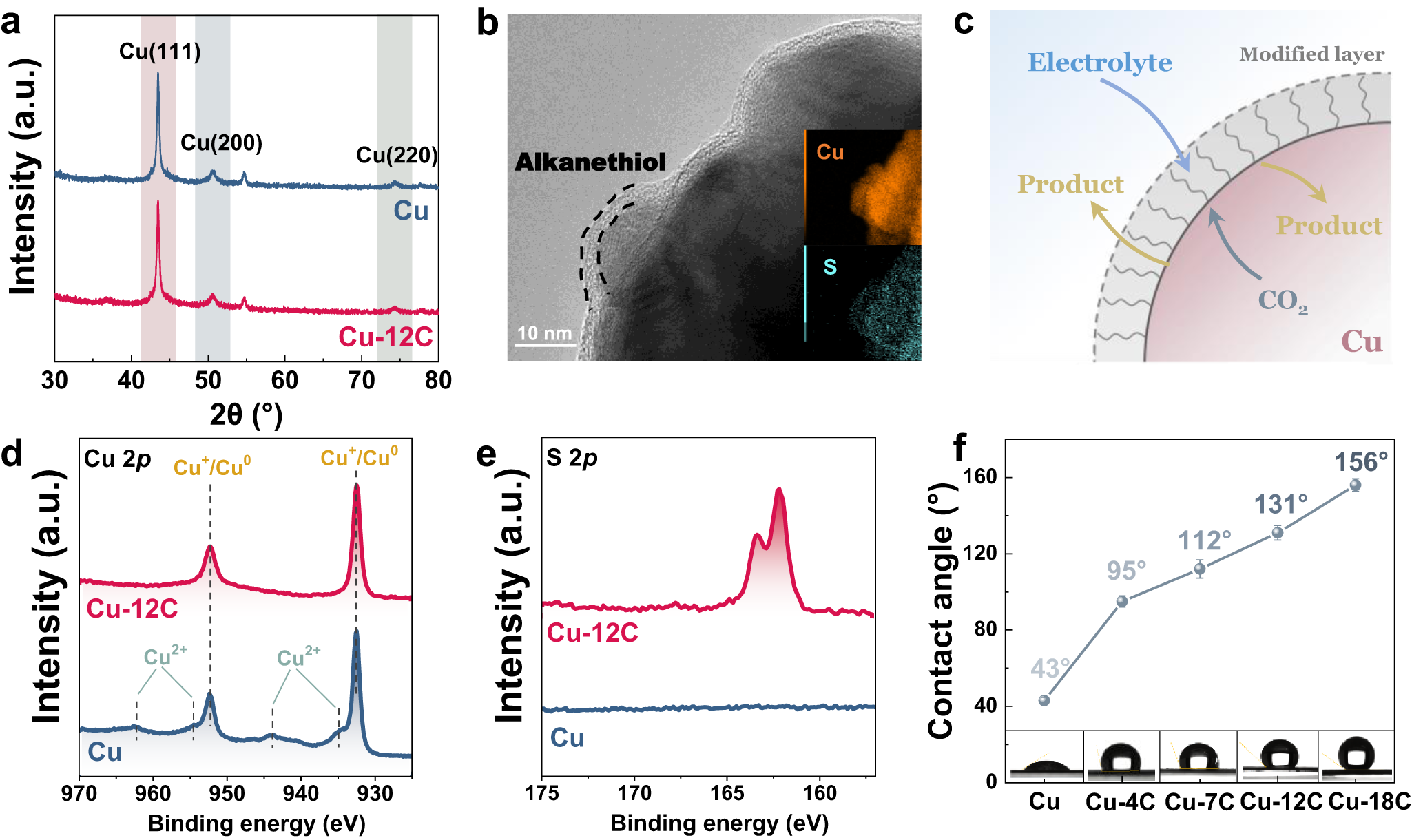
Tunable CO2 electroreduction to ethanol and ethylene with controllable interfacial wettability

Advanced Photon Source University of Chicago News

A review on CO2 hydrogenation to ethanol: Reaction mechanism and experimental studies - ScienceDirect

Nanospikes Convert Carbon Dioxide Back Into Ethanol
Chemistry University of Chicago News
Chemistry University of Chicago News
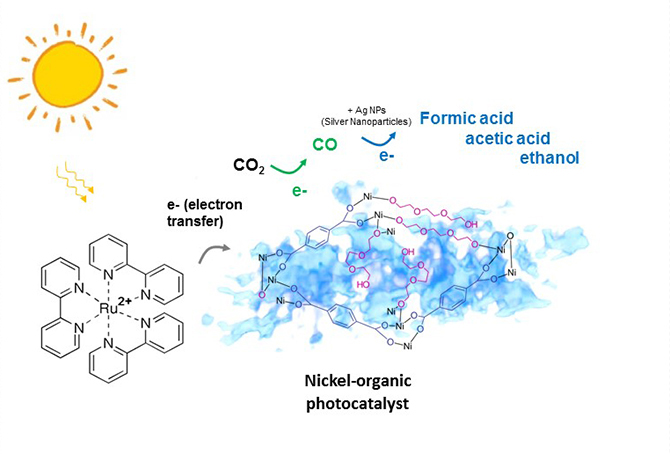
New light-activated catalyst grabs CO2 to make ingredients for fuel







