Researchers discover a way to tease oxygen molecules from carbon
() —A small team of researchers with the University of California has found a way break apart carbon dioxide molecules and get carbon atoms and oxygen molecules instead of carbon monoxide and an oxygen atom. In their paper published in the journal Science, the team describes how they did it, and the implications of their findings. Arthur Suits and David Parker offer a perspective piece in the same journal issue that describes in more depth, minimum energy path (MEP) where reactants don
() —A small team of researchers with the University of California has found a way break apart carbon dioxide molecules and get carbon atoms and oxygen molecules instead of carbon monoxide and an oxygen atom. In their paper published in the journal Science, the team describes how they did it, and the implications of their findings. Arthur Suits and David Parker offer a perspective piece in the same journal issue that describes in more depth, minimum energy path (MEP) where reactants don't always follow the easiest path during chemical reactions and how it pertains to the work done by this group.

Detected: sulfur compound created by photochemistry in exoplanet
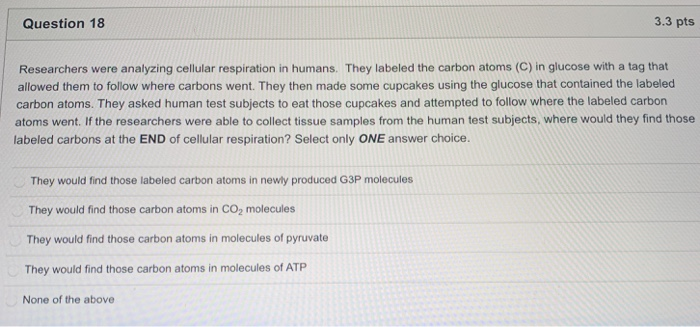
Solved Question 18 3.3 pts Researchers were analyzing

Quick, easy and early diagnosis with rare earth ions
Can we build a device that converts carbon dioxide to oxygen? If

The Ocean's Tiny Chemists – Woods Hole Oceanographic Institution
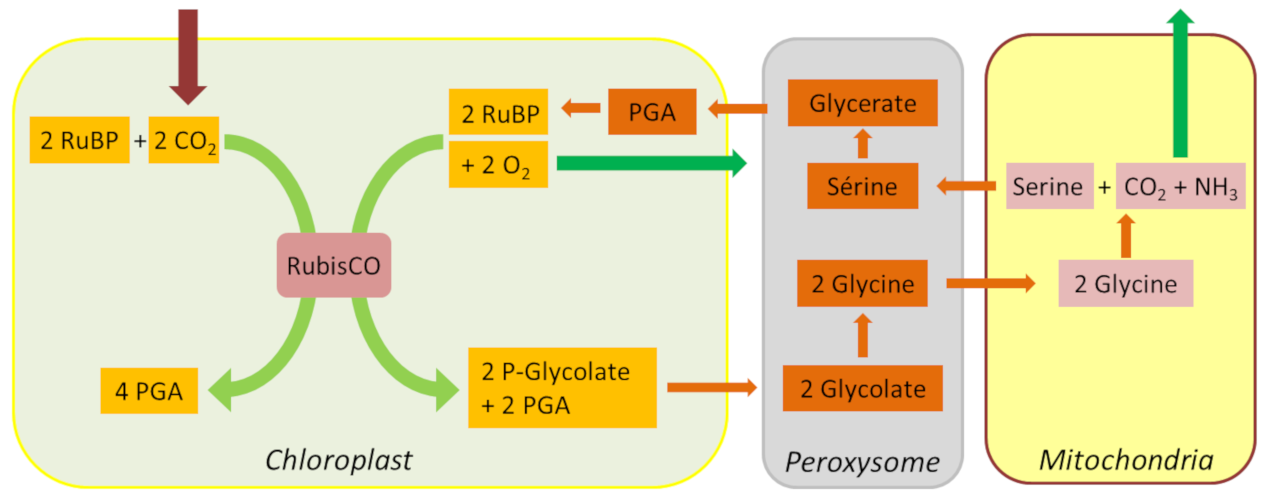
The path of carbon in photosynthesis - Encyclopedia of the Environment

Researchers discover unique property of critical methane-producing

A proposal for vacuum ultraviolet torch using plasmonic-based

New ceramic bonding technique promises rigid composite structures

Why stressed-out corals lose their color: Scientists find new clues
How can we separate carbon dioxide to carbon and oxygen like

New ceramic bonding technique promises rigid composite structures

Bad Astronomy, NASA's MOXIE made oxygen on Mars
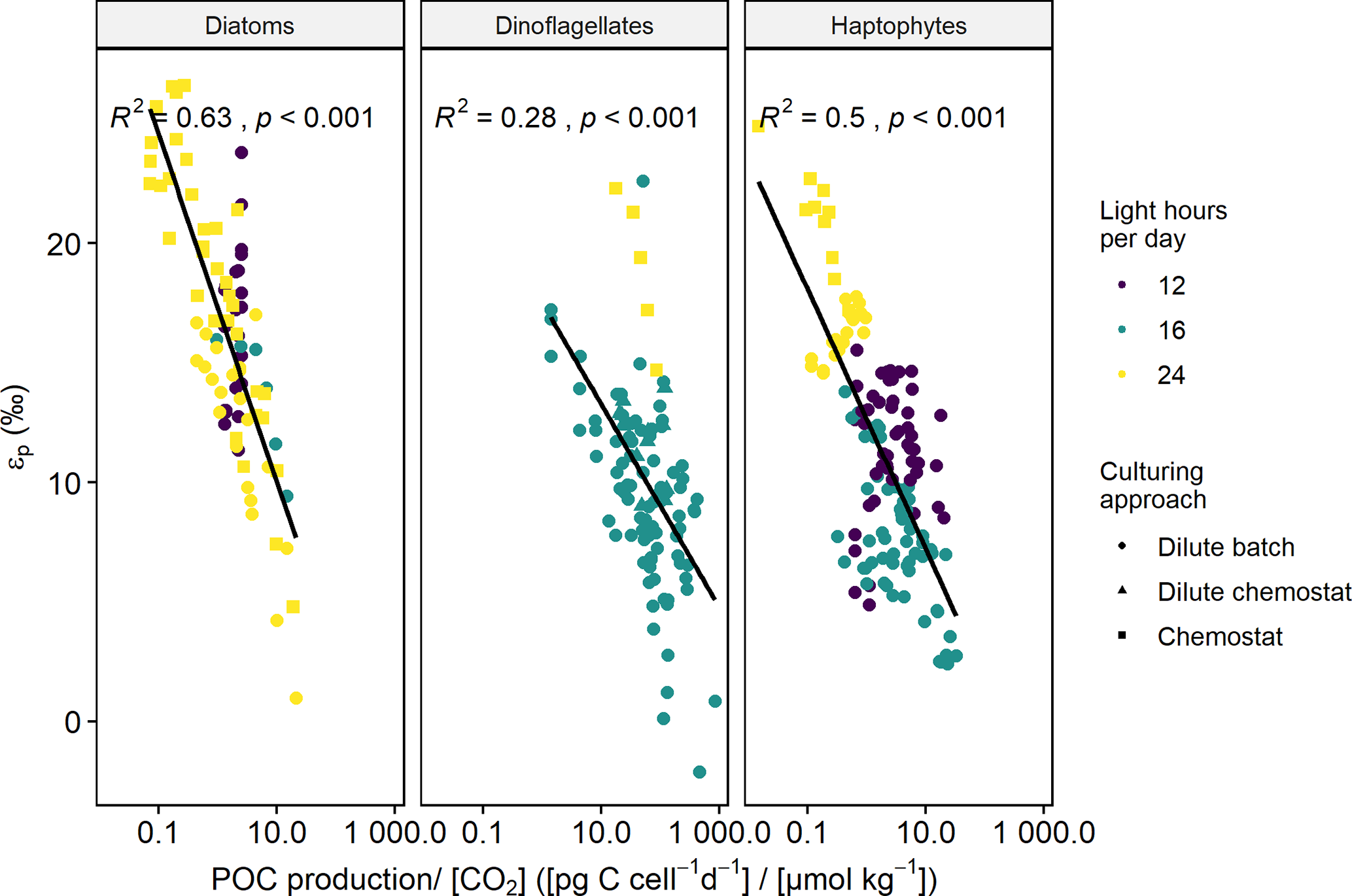
BG - Physiological control on carbon isotope fractionation in

New ceramic bonding technique promises rigid composite structures