An ideal gas is taken from (Pi , Vi ) to (Pi , Vi ) in three different ways. - Sarthaks eConnect
An ideal gas is taken from (Pi , Vi ) to (Pi , Vi ) in three different ways. Identify the process in (d) Equal work is done in Process A, B & C

One mole of an ideal gas is taken from state A to state B by three different processes a A C B, b A D B and c A E B as
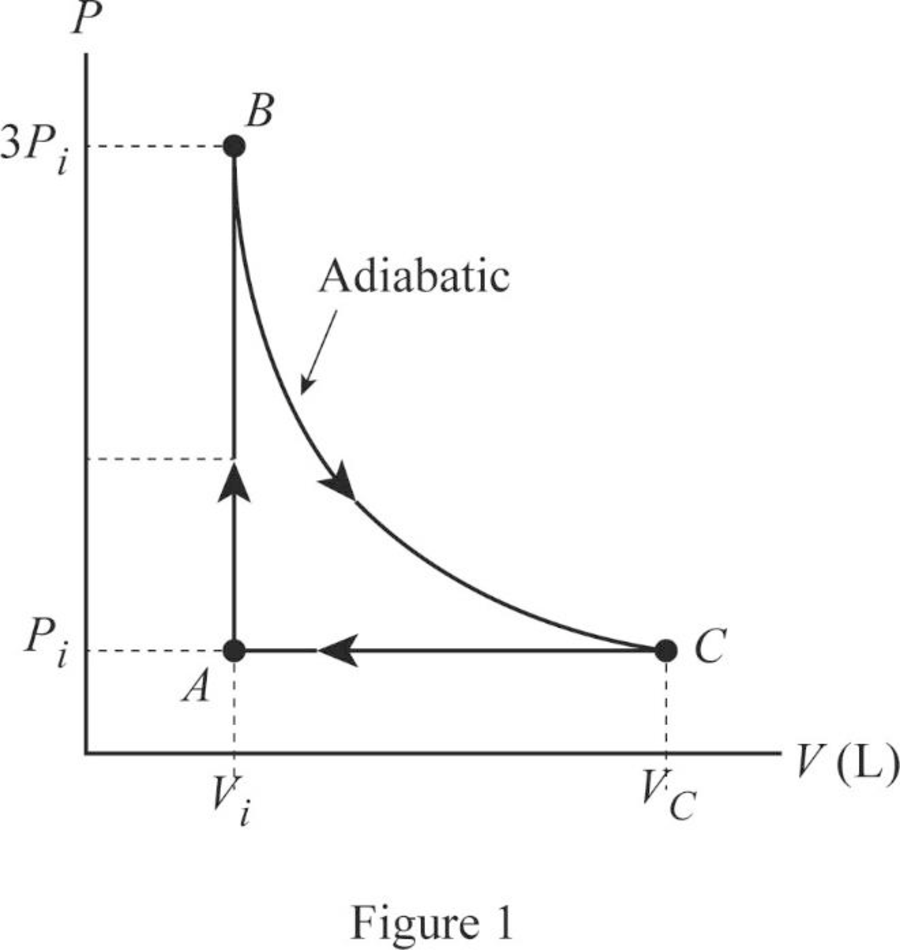
An ideal gas with specific heat ratio γ confined to a cylinder is put through a closed cycle. Initially, the gas is at P i , V i , and T i .

An ideal gas initially at Pi, Vi, and Ti, is taken through a cycle as shown below. (Let the factor n= 3.1.) a) Find the work done on the gas per cycle

Solved An ideal gas initially at Pi, Vi, and Ti is taken

Solved An ideal gas initially at Pi, Vi, and T is taken

Solved A system of ideal gas can be taken from state 3 to

An ideal gas is taken around ABCA as shown in the above P-V diagram. The work done during a cycle is.2pVpVdfrac{1}{2}pVzero

An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most. - Physics

An ideal gas initially P_i ,V_i , and T_i is taken through a cycle as shown in Figure. (a) Find the net work done on the gas per cycle 1.00 mol of
2 moles of an ideal gas undergoes isothermal compression along three different paths - Sarthaks eConnect

etwork cone zus ? / 3x1 Fix Skaws (P-V) diagram of an ideal gas under goes chance from A -> B, in the different process (1) In which work done is Maximum

An ideal gas is taken from state A to state B via three different processes as shown in the pressure volume (P-V) diagram. If Q4, Q, & Q, indicates the heat absorbed
An ideal gas is taken from (Pi , Vi ) to (Pi , Vi ) in three different ways. - Sarthaks eConnect

An ideal gas is taken around the cycle ABCA shown in P - V diagram. The net work done by the gas
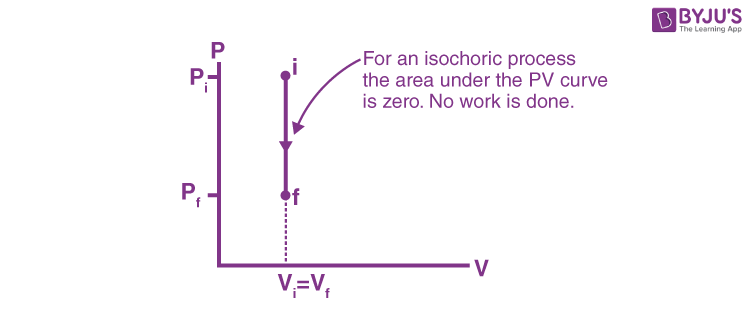
Isochoric Process - Definition, Example, Formula, P-V Diagram





:strip_icc()/reena.sotropa.-098ff275145c42f1a5aca1ff979ad355.jpg)



:strip_icc()/reena.sotropa.-098ff275145c42f1a5aca1ff979ad355.jpg)

