PDF] Two Simple yet Accurate Equations for Calculating the Fugacity Coefficient Phi and the Gas Compressibility Factor

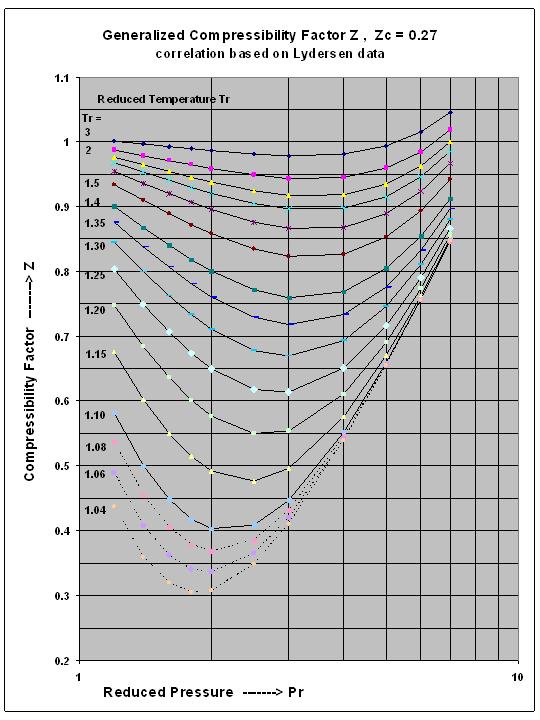
Two Simple yet Accurate Equations for Calculating the Fugacity Coefficient Phi and the Gas Compressibility Factor Z . by A.E. for mychemengmusings.wordpress.com In this post two new powerful equations are presented one for the Gas Compressibility Factor ‘Z’ and one for the Gas Fugacity Coefficient ‘phi’. Both give excellent prediction results for the sub-critical reduced pressure region and superheated vapor region. These two equations are surprisingly simple, allow direct calculation without the need for iterations hence easy to implement in spreadsheets or used on handheld devices and calculators! The two form a thermodynamically consistent pair. Three Charts have been prepared mapping out the predictions made with these equations. Numerical calculation examples are given including for superheated Steam, Ethane, Propane and Propylene. The basis for each of these equations is presented in Part III of this post giving ample attention to the basis on which these equations rest and their validation against measured data.

PDF] Two Simple yet Accurate Equations for Calculating the Fugacity Coefficient Phi and the Gas Compressibility Factor

B49CE Student Guide v1 PDF, PDF, Gibbs Free Energy
The fugacity of a gas is greater than its pressure. What can be said about the gas internal pressure? - Quora

A Robust Algorithm for Roots Selection and Saturation Pressure Calculation for Cubic Equations of State

DOC) Computation of the Compression Factor and Fugacity Coefficient of Real Gases

PDF) Calculating the vapor-liquid phase equilibrium for multicomponent systems using the Soave-Redlich-Kwong equation

Compressibility Factor Z for sub-critical pressures for Lee-Kesler's “Simple, Normal Fluids” Z-LK with a new set of equations for Excel Spreadsheets

Modeling of CO2 Freeze-Out in the Processing of CO2-Rich Natural Gas

Two Simple yet Accurate Equations for Calculating the Fugacity Coefficient Phi and the Gas Compressibility Factor Z

PDF) Accuracy of Approximations to the Poynting Correction for Ice and Liquid Water